Research
Epigenetic Control of Oocyte Quality and Early Embryo Development
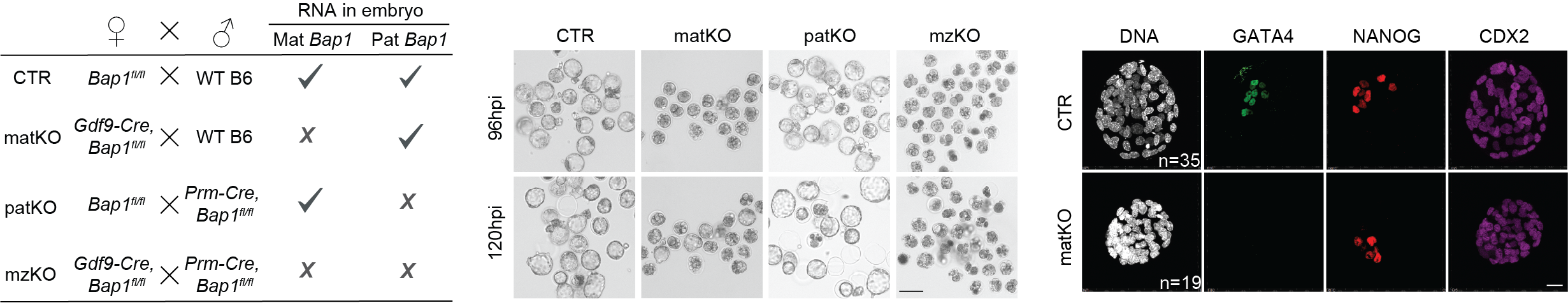
The oocyte is among the most epigenetically specialized cells in the body, poised to support the development of a new life upon maturation. Rather than a blank state, it establishes and transmits a distinct chromatin landscape that instructs the embryo which genes to activate, which to silence, and how to organize its genome during the earliest stages of life.
Our laboratory investigates how this chromatin state is established, maintained, and reprogrammed, and how its disruption leads to infertility and embryo failure. We study this process across the full developmental trajectory — from the establishment of histone modification landscapes during oocyte growth to their remodeling in the preimplantation embryo and their role in shaping zygotic genome activation and early cell fate decisions.
By combining mouse genetics, ultra-low-input chromatin profiling, and single-embryo transcriptomics, we define the epigenetic logic of early development. A central focus is how the balance between chromatin activation and repression is controlled in the oocyte, including mechanisms that safeguard developmental competence and enable the maternal-to-zygotic transition.
Transposable Elements in Totipotency and Zygotic Genome Activation
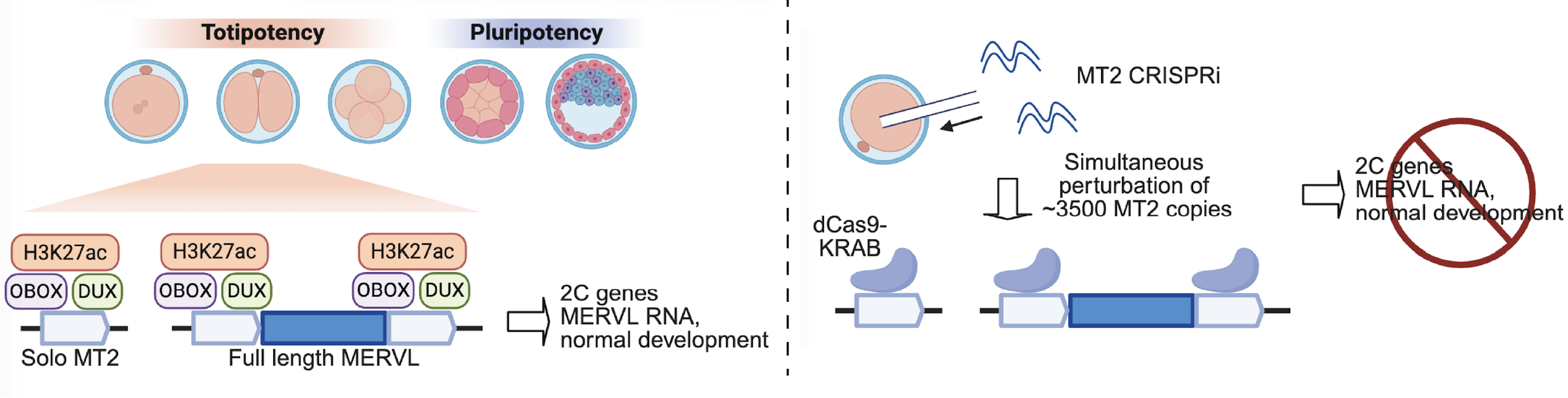
Zygotic genome activation (ZGA) marks the moment when the embryo first executes its developmental program. A striking feature of this transition is the transient activation of retrotransposons, which are otherwise generally silenced despite comprising a large fraction of the genome.
Our laboratory investigates how these elements are integrated into the regulatory architecture of early embryos. We study how specific retrotransposon families function as cis-regulatory elements, acting as promoters and enhancers that drive the ZGA transcriptional program and contribute to the establishment of totipotency.
Using perturbation approaches in mouse embryos and extending to human stem cell models, we examine how retrotransposon activation and silencing are precisely controlled, and how their misregulation leads to developmental failure. This work connects the regulatory logic of ZGA to early embryo viability, including failure at the onset of genome activation.